TP53 Point Mutation, from molecular mechanisms to therapeutic strategies

The TP53 gene, also known as the p53 gene, is one of the most critical tumor suppressor genes in the human genome. Discovered in 1979 and recognized for its tumor-suppressive function in 1989, TP53 has since been regarded as the “guardian of the genome” and remains a central focus of cancer research.
Under normal physiological conditions, the TP53 gene functions as a tightly regulated stress-response hub . The p53 protein encoded by the TP53 gene is constantly produced but also rapidly degraded. Upon DNA damage, replication stress, or metabolic imbalance, p53 becomes stabilized and activated, allowing it to orchestrate cell fate decisions.
Rather than simply inducing apoptosis, p53 exerts a context-dependent regulatory role:
- In response to mild damage, p53 induces cell cycle arrest through transcriptional activation of targets such as p21, allowing time for DNA repair.
- Under moderate stress, p53 promotes DNA repair pathways and antioxidant responses.
- When damage is irreversible, p53 triggers apoptosis or cellular senescence to eliminate potentially malignant cells.
This context-dependent regulatory capacity makes the TP53 gene a central guardian of genomic stability.
In experimental studies, TP53 antibodies are widely used to assess p53 protein expression, conformational changes, and mutation-associated alterations, serving as essential tools for dissecting TP53 signaling activity and the functional consequences of TP53 mutations.
TP53 Point Mutations and Biological Consequences
When TP53 gene mutations occur, this finely tuned regulatory system collapses. In human cancers, TP53 mutations are predominantly point mutations, particularly missense mutations. These alterations can arise as:
- Somatic TP53 mutations, acquired during tumor development;
- Germline TP53 gene mutations, which are inherited and associated with Li-Fraumeni syndrome.
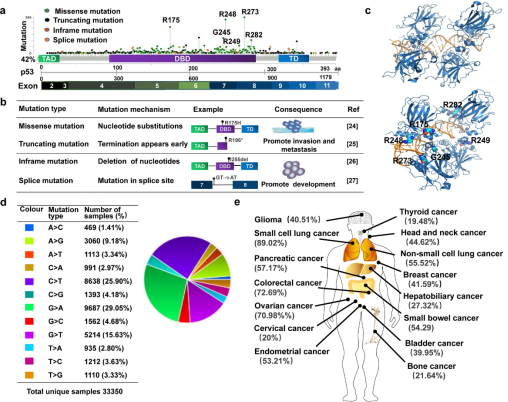
Most TP53 point mutations occur within the DNA-binding domain, leading to conformational changes in the p53 protein. As a result, mutant p53 may exhibit loss-of-function (LOF) or gain-of-function (GOF) properties, actively promoting tumorigenesis.The biological consequences of TP53 point mutations include:
- Increased cancer susceptibility, with germline TP53 mutation carriers showing a lifetime cancer risk exceeding 90%;
- Poor prognosis and therapy resistance, as mutant p53 tumors often display aggressive behavior and reduced sensitivity to radiotherapy and chemotherapy;
- System-level effects, where mutant p53 reshapes the tumor microenvironment, enhances angiogenesis, and facilitates immune evasion.
Therapeutic Strategies Targeting TP53 Mutations
Unlike kinase-driven oncogenic mutations, targeting TP53 mutations remains challenging due to the complexity of the p53 signaling network and the heterogeneity of TP53 gene mutations. Current research directions include:
In addition, tumors harboring TP53 mutations often exhibit a high mutational burden, making them more responsive to immunotherapies such as immune checkpoint inhibitors.
EDITGENE has established a comprehensive portfolio of TP53 gene-edited cell lines, including TP53 point mutation and TP53 knockout models. These standardized and functionally validated cell models support mechanistic studies, drug discovery, and translational cancer research.


Leveraging the newly developed Bingo™ Platform , EDITGENE applies an optimized Prime Editing (PE7 system) to introduce precise point mutations with editing efficiencies exceeding 90% at the target locus.
This advanced gene-editing strategy enables the generation of high-efficiency, genetically stable point mutation cell lines, all of which are molecularly validated and suitable for basic research and drug discovery applications.
References
1. Chen X, Zhang T, Su W, Dou Z, Zhao D, Jin X, Lei H, Wang J, Xie X, Cheng B, Li Q, Zhang H, Di C. Mutant p53 in cancer: from molecular mechanism to therapeutic modulation. Cell Death Dis. 2022 Nov 18;13(11):974.


Comments
Post a Comment